
When you hear about generic medicines, you might picture an exact chemical copy of a brand-name pill. But when we talk about biological medicines, specifically monoclonal antibody biosimilars, the rules change completely. These aren't simple copies. They are complex biological products that mimic the behavior of an original medicine, often called the reference product, without being chemically identical down to the atom. As of 2026, this distinction remains the most critical detail for both clinicians and patients to understand before starting treatment.
The landscape has shifted dramatically since the first monoclonal antibody biosimilar gained worldwide approval back in 2013. Today, these treatments are essential tools in fighting cancer and autoimmune diseases, offering significant cost advantages while maintaining high safety standards. Let's break down exactly how they work, which ones are available right now, and how they differ from standard generics.
Understanding Biological Similarity vs. Chemical Identity
To grasp why biosimilars are unique, we need to look at what makes them different from traditional small-molecule drugs. A common antibiotic is like a Lego brick-you can build an identical copy easily. But a monoclonal antibody is more like a massive protein structure. It has a molecular weight of approximately 150,000 daltons. For comparison, insulin sits around 5,808 daltons. Because of this sheer size and complexity, manufacturing processes naturally create small variations in the protein molecule.
| Feature | Small-Molecule Generic | Biosimilar |
|---|---|---|
| Molecular Weight | Low (<1,000 Da) | High (~150,000 Da) |
| Manufacturing | Chemical synthesis | Living cells |
| Approval Requirement | Chemical equivalence | No clinically meaningful differences |
That variation is key. The U.S. Food and Drug Administration (FDA) defines these products as highly similar to the reference product with no clinically meaningful differences in safety, purity, and potency. This means the tiny structural differences shouldn't impact how the body reacts to the drug. However, achieving this requires rigorous testing, particularly regarding post-translational modifications like glycosylation patterns. In the past, minor changes here have caused issues, such as the alpha-1,3-galactose epitope reactions seen with certain cetuximab formulations, so modern development focuses heavily on matching these sugar-chain structures precisely.
Regulatory Pathways and Global Approvals
The journey to get these medicines on the shelf involves strict oversight. The European Medicines Agency (EMA) laid the groundwork with their draft guidelines released on November 18, 2010. Shortly after, the United States followed suit. On March 6, 2015, the FDA approved the first biosimilar product in the U.S., filgrastim-sndz (marketed as Zarxio by Sandoz).
Fast forward to 2026, and the numbers reflect robust industry growth. As of 2023, the EMA had granted marketing authorizations for over 50 biosimilars since 2006, with about 35% of those representing monoclonal antibody therapies. The FDA has also been aggressive in clearing new candidates. This regulatory momentum ensures that every new approval undergoes extensive comparative analytical, non-clinical, and clinical programs to establish similarity. Manufacturers must conduct studies to identify the most sensitive clinical indication for detecting potential differences, ensuring that even subtle shifts in efficacy are caught.

Approved Monoclonal Antibody Biosimilar Examples
Knowing which specific drugs are available is crucial for understanding the current market options. Several blockbuster originators have already reached the stage where multiple biosimilar competitors exist. Here are the primary examples currently shaping oncology and immunology practices.
- Bevacizumab (Avastin): Used for colorectal cancer and glioblastoma. As of late 2023, six biosimilars were approved, including Mvasi, Zirabev, Alymsys, Vegzelma, Avzivi, and Jobevne.
- Rituximab (Rituxan): Essential for non-Hodgkin's lymphoma and chronic lymphocytic leukemia. Three biosimilars made it through US approval: Truxima, Ruxience, and Riabni.
- Trastuzumab (Herceptin): Critical for HER2-positive breast cancer. Six versions are approved in the US, such as Ogivri, Herzuma, Ontruzant, Trazimera, Kanjinti, and Hercessi.
Beyond these heavy hitters, other categories are expanding. We see six filgrastim biosimilars preventing chemotherapy-induced neutropenia (like Fulphila and Ziextenzo) and Retacrit for anemia management. These listings demonstrate that the therapy is no longer theoretical; it is a tangible reality in hospital pharmacies.
| Reference Product | Biosimilar Brand Name | Approx. Approval Date |
|---|---|---|
| Bevacizumab | Mvasi | September 14, 2017 |
| Rituximab | Truxima | November 28, 2018 |
| Trastuzumab | Ogivri | December 1, 2017 |
| Infliximab | Remsima | July 21, 2023 (Interchangeable) |
One standout moment in recent history occurred on July 21, 2023, when Celltrion's Remsima became the first monoclonal antibody biosimilar designated as "interchangeable" by the FDA. This designation allows pharmacists to switch the medication without needing a new prescription from the doctor, much like standard generics, provided state laws allow it. This step significantly streamlines the logistics for clinics managing high volumes of patients.
Clinical Adoption and Cost Effectiveness
Why does this matter to a patient or provider? Cost and access. A major barrier to advanced biologic therapy has historically been price. Biosimilars address this directly. In a 2022 study published in JAMA Oncology, researchers tracked patients switching from reference rituximab to the biosimilar Truxima. Across 1,247 patients at 15 US cancer centers, the switch resulted in an average cost reduction of 28% per treatment cycle. Crucially, there were no significant differences in effectiveness or safety outcomes.
Industry analysts project that by 2027, monoclonal antibody biosimilars could capture a substantial portion of the market. Evaluate Pharma estimated in June 2023 that these products would account for 45-65% of the market share of originator products within three years of launch. With cumulative savings projected at $250 billion in the US healthcare system between 2023 and 2028, the financial incentive for adoption is massive. Cancer therapies alone make up 62% of the prescription volume for this class.

Safety Profiles and Immunogenicity Concerns
While costs are lower, safety remains non-negotiable. One persistent concern is immunogenicity-where the body creates antibodies against the medicine itself, reducing its effect or causing allergic reactions. The EMA's 2021 safety report documented 12 cases of unexpected immune responses across 1.2 million patient-years of exposure. That rate (0.001%) is statistically equivalent to the reference products. This data reassures providers that the risk profile hasn't changed simply because the manufacturer did.
Hurdles remain, however. Patent litigation is frequent, with an average of nearly 15 challenges per product. Additionally, pharmacy benefit managers sometimes restrict formularies, affecting 32% of launches. Providers also face educational gaps; a survey showed only 58% of oncologists felt "very confident" prescribing biosimilars in 2022. Yet, as more real-world evidence accumulates and training improves, confidence levels are expected to rise alongside the availability of newer generations like adalimumab (Humira) and pembrolizumab (Keytruda) biosimilars entering the pipeline.
Frequently Asked Questions
Are biosimilars exactly the same as the original drug?
No, they are highly similar but not exact chemical copies due to the complex nature of biological molecules produced by living cells. Regulatory bodies require them to show no clinically meaningful differences in safety and efficacy, meaning they perform the same way in your body despite minor structural variances.
What is an interchangeable biosimilar?
This is a special designation requiring proof that switching between the reference product and the biosimilar causes no additional risks. As of 2023, Remsima became the first monoclonal antibody with this status, allowing pharmacist substitution without doctor intervention in many jurisdictions.
Can I switch between my brand drug and a biosimilar?
For products designated as interchangeable, switching is generally safe and often automatic via pharmacy formulary changes. For others, it usually requires a new prescription and monitoring by your healthcare provider to ensure your response remains stable.
Do biosimilars work as well for cancer treatment?
Yes. Studies like the one in JAMA Oncology comparing rituximab biosimilars showed comparable effectiveness in treating cancers like lymphoma. They maintain the necessary potency to target specific cancer markers effectively.
Why are biosimilars cheaper than the reference product?
Because biosimilar manufacturers do not incur the initial costs of discovering the molecule or conducting the full suite of early clinical trials needed for the originator. This saves money and competition drives prices down, though quality controls remain strictly enforced.
14 Comments
Write a comment
More Articles

Understanding Eye Cancer and How Positive Thinking Helps
Learn what eye cancer is, its main types, and how a positive mindset can improve treatment outcomes and quality of life.

Crohn's Disease and Gluten Intolerance: Key Signs and What to Watch For
Explore the link between Crohn's disease and gluten intolerance, learn warning signs, testing methods, and dietary steps to manage symptoms effectively.
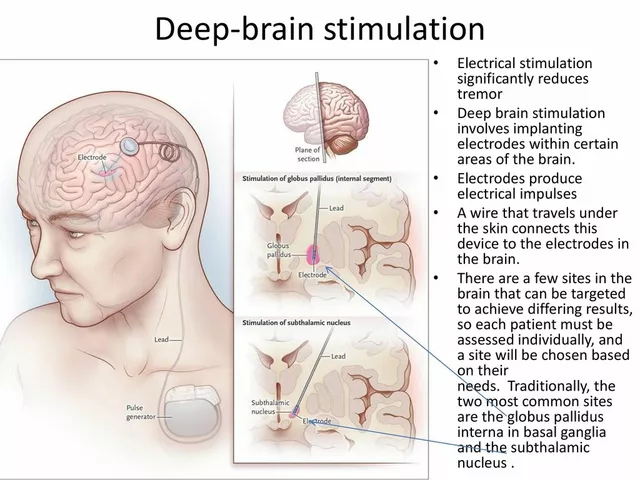
Deep Brain Stimulation for Tremor Management: A Comprehensive Guide
In my latest blog post, I've created a comprehensive guide on Deep Brain Stimulation (DBS) for tremor management. This innovative treatment has been proven to effectively reduce tremors in patients with Parkinson's disease and essential tremor. Through a surgical procedure, a device is implanted to send electrical signals to specific areas of the brain, offering relief to those struggling with involuntary shaking. I've included detailed information on the procedure, its benefits, and potential risks to help you better understand this life-changing treatment option. If you or a loved one are experiencing tremors, I highly recommend giving it a read to see if DBS could be the right solution for you.

Sabrina Herciu
March 29, 2026 AT 11:47It really matters when you look at the cost breakdown! The studies show significant savings! We must consider the long term effects carefully.. And also the manufacturing differences matter too! Some people forget the glycosylation patterns entirely.. That is a huge deal for the immune system function..!! The FDA requires strict testing protocols always.. Don't ignore the data released in 2023..!! The market share projections are quite interesting too.. Five hundred billion dollars in savings is significant globally..!! You cannot underestimate the impact on healthcare systems.. Clinicians need better training before prescribing freely..!! Education is key for higher adoption rates overall..!! Patients deserve complete transparency about switching drugs..!! Interchangeability helps pharmacists act much faster..!! But state laws vary widely across the entire nation..!! Safety monitoring remains essential after initial approval..!! Immunogenicity rates stay low according to recent reports..!! Everyone benefits when medical costs drop significantly..!! We should always support evidence-based medicine decisions..!!